Marine "Ich" or "White Spot" is probably the most frequent fish disorder encountered by aquarists keeping tropical marine fish. It is also probably the disorder that is most misunderstood by aquarists. However, because this disorder is also encountered in aquaculture, substantial research has been undertaken and the picture is somewhat clearer than many believe.
Marine "Ich" is caused by a ciliated protozoan called Cryptocaryon irritans Brown 1951. Ciliates (Phylum Ciliophora) are one of the largest groups of protozoans and all posses cilia or compound ciliary structures for food acquisition or locomotion at some point in their life cycle. Of the some 7200 species that have been described, around one third are ecto- and endo- commensals and parasites (Ruppert and Barnes, 1994). C. irritans is an obligate ectoparasite (Dickerson and Clark, 1996) which means that it is an external parasite that needs the fish host to complete its life cycle. The name 'Marine "Ich"' has been coined because it is essentially the marine equivalent on Ichthyophthirius multifiliis Fouquet, 1876, or "Ich" for short. While the two species of ciliates have superficially similar life cycles and signs, they have been found to be only distantly related (Wright and Colorni, 2002) .
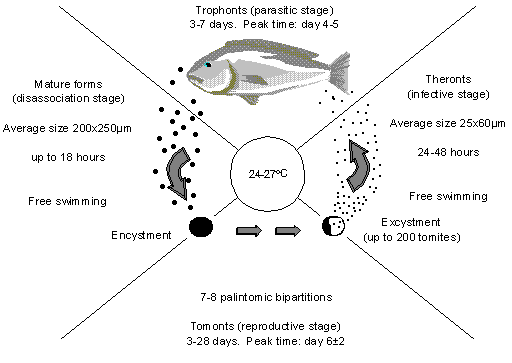
C. irritans has a four stage life cycle, as shown in Figure 1. The parasitic stage (trophonts) is the one that results in the appearance of white spots all over the fish. The trophonts burrow under the skin where they feed on body fluids and tissue debris. When the trophonts first infect the fish they are small but grow as they feed and so the white spots are initially small but get larger as they mature. Once mature, they drop off the fish and sink/swim down to the substrate where they encyst and begin to reproduce. In this stage they are called tomonts. After a number of days in which the tomonts divide, the cyst ruptures, releasing the tomites. Tomites may differentiate into theronts, the infective stage, which actively seek a host to reinfect.

Figure 1: The life cycle of C. irritans (After Colorni, 1987).
When the theronts (parasitic stage) are embedded in the skin of the fish, the fish secrete a thick layer of mucus in response to the irritation and they are protected from outside influences. This makes treatment of infected fish difficult.
Table 1 shows the lengths of each of the stages of C. irritans as determined by various authors and studies. Despite each of the authors working in different locations and having different sources for the parasites, the findings are consistent. Trophonts will attach to the fish for at least 3 days (78 hours) and as long as 7 days at around 25°C. When the trophonts drop off the fish, they can take as little as 30 minutes and up to 24 hours to reach the substrate, with the majority encysting towards the shorter end of the range. The reproductive stage appears to be quite variable. Reproduction can be complete in as little as 84 hours (3 days) but can extend to as long as 12 days and even longer.
Table 1: Lengths of stages of C. irritans from various studies.
| Reference | Trophont | Free-swimming Trophont | Tomont | Theront | Temperature |
| Burgess and Matthews, 1994a | 78 - 113 hrs | 2 - 8 hrs | 84 hrs - 35 days | up to 18 hrs | 23 - 27°C |
| Burgess and Matthews, 1994b | 78 - 113 hrs | 10 - 12 days | 24 - 26°C | ||
| Cheung et al., 1979 | 30 mins - 24+ hours | 8 - 9 days | 25°C | ||
| Cheung et al., 1979 | 30 mins - 24+ hours | 5 - 7 days | 30°C | ||
| Colorni, 1985 | 3 - 7 days | up to 18 hours | 3 - 28 days | 24 - 48 hours | 24 - 27°C |
| Diggles and Lester, 1996a | 3 - 7 days | 3 - 12 days | 25°C | ||
| Diggles and Lester, 1996b | 4 - 6 days | 3 - 15 days | 25°C | ||
| Yoshinaga and Dickerson, 1994 | 6 - 7 days | 4 - ? days | up to 12.5 hours | 23 - 25°C |
The first sign of Marine "Ich" infections are white spots. The fish develop small white cysts on their body surfaces (Figure 2). The eyes may also become infected causing clouding of the eyes and even blindness. Fish may also scratch, show a loss of appetite and show laboured breathing, especially when the gills are infected. Some fish may even jump out of the tank.


Figure 2 a & b: Chaetodon guentheri with severe C. irritams infection.
As the integument of the fish is often compromised, particularly with severe infections, the fish may suffer degredation or even loss of osmotic control.
Innate immunity refers to the general response to an invading pathogen or parasite regardless of that pathogen or parasite encountered (Dickerson and Clark, 1996). This form of immunity does not rely on previous encounters and includes generalised reactions such as secretion of mucus, but may include specific host cell responses (acquired genetically).
While little formal study has been performed on innate immunity of marine fish to C. irritans, innate immunity of freshwater fish to I. multifiliis, both between and within host species suggests that the former may be possible. Collective anecdotal evidence from marine aquarists lends weight to the idea that some species, such as chaetodontids (butterflyfish) and acanthurids (surgeonfish and tangs) may be more prone to Marine "Ich" infections, whereas other species such as callionymids (dragonets) are not at all. Intraspecific differences in innate immunity would be much harder to detect through random observation.
Acquired immunity occurs when the response is specific to the invading organism, which is recognised directly or through antigens (Dickerson and Clarke, 1996). Colorni (1987) first suggested that marine fish could acquire some immunity to C. irritans by surviving several infections. Burgess and Matthews (1995) demonstrated acquired immunity in the thick-lipped mullet, Chelon labrosus. They found that 82% of fish that had been previously exposed to high levels of theronts were immune to a secondary exposure.
There are only two proven methods for the treatment of Marine "Ich", copper and hyposalinity. Neither method can be used in the presence of elasmobranchs (sharks and rays) or invertebrates and so treatment must be performed in a quarantine or hospital tank. Hyposalinity is the preferred treatment as it is not dangerous to the fish and actually eases osmotic stress on the fish. Hyposalinity can also be performed in the presence of calcareous substrates.
For a more detailed description of hyposalinity, please see: Hyposalinity.
Copper is toxic to both fish and invertebrates and must be used with care. As calcium carbonate absorbs copper, copper cannot be used in tanks with calcareous substrates, such as coral sand or shell grit, or with coral decorations. Copper should not be used in the presence of invertebrates or elasmobranchs. Copper is not very stable in sea water and the levels must be continuously monitored.
Other methods for the treatment of marine "Ich" have been described, but, generally, they limited success. One of the problems with assessing treatment methods for marine "Ich" is that fish may recover through acquired immunity giving the impression that the treatment was effective when perhaps the fish would have recovered on their own.
It is worth noting that many people have had apparent success with adding garlic to the food of infected fish. Garlic is well know for its therapeutic effect, particularly in humans and it is possible that the same active ingredients may be effective in fish, too. As with other treatments, it is difficult to assess the effectiveness of garlic as a dietary supplement. The theory behind the use of garlic is to aid and strengthen the fish's immune system's ability to recognise and react to the parasite.
Prophylactic use of copper or hyposalinity in a quarantine tank is the only sure way of preventing the introduction of C. irritans parasites into a display tank. However, generally, these drastic measures are not necessary and simply keeping a fish for 6 weeks in a quarantine tank is ample time for signs of the parasite, if present to become evident. Further, copper is very toxic and may weaken or kill the fish if not used with care.
As previously mentioned, marine "Ich" is generally misunderstood by aquarists. Many myths that abound about its causes, prevention and treatment.
Many aquarists regard common cleaner wrasses, Labroides dimidiatus, as effective agents for the control and treatment of marine "Ich". While this would be a desirable situation, it is unfortunately not true. Grutter (2000) showed that L. dimidiatus feeds mainly on the larvae of gnatid isopods throughout its life, and C. irritans theronts were never found in it's diet. Fish remain infected with theronts despite the presence of cleaner wrasse, and even the cleaner wrasse may become infected (personal observation).
Freshwater dips are generally ineffective in the treatment of marine "Ich" infestations. Trophonts burrow deeply into the epithelium where they are generally protected from external influences. Colorni (1985) found that even after 18 hours in freshwater, infected fish still have trophonts attached in the same positions as they had held before the freshwater treatment. The trophonts later detached and completed their life cycle as normal.
Trophonts penetrate the epithelium which causes a loss in osmoregulatory capability. Infected fish are less able to cope with a sudden and drastic change in the ionic concentration of their environment. This is likely to cause further stress to the fish which will impair their ability to acquire immunity to the parasite.
A specific gravity of 1.017 is not effective in controlling outbreaks of marine "Ich" as has been claimed by some authors (e.g. Delbeek and Sprung, 1994). Colorni (1985) found that tomonts could survive at salinities as low as 15 ppt (specific gravity ~1.011 d20/20). Further, a specific gravity of 1.017 is far too low for most invertebrates and the long term effect on fish is unknown.
There is a widely held belief in the marine aquarium hobby that "Ich" is always present in our aquaria and this belief is often repeated on marine bulletin boards. There is much information in the scientific literature that contradicts this belief.
C. irritans is an obligate parasite (Burgess and Matthews, 1994; Dickerson and Dawe, 1995; Yoshinaga and Dickerson, 1994). Obligate means the parasite can not survive without infecting its host, in this case, fish. Theronts have been shown to die if a suitable host is not found within the required time. Yoshinaga and Dickerson (1994) found that few theronts (0.34%) were viable 12.5 hours after excystment and Burgess and Matthews (1994) found that no theronts were viable 18 hours after excystment. Colorni (1985) found that some excysted tomites (=theronts) were observed to be moving weekly after 48 hours. While the life span of the theronts appears variable, it is limited and all will die without finding a suitable host.
If an aquarium has no fish in it, and there are no additions of fish, or anything else that could be carrying trophonts, tomonts, tomites or theronts for a period of 6 weeks or longer, all parasites will have died. An aquarium such as this is an obvious exception to "Ich" always being present.
Many fish collected for marine aquariums will not be carrying "Ich". Incidence of C. irritans in wild fish varies widely and may be geographically related. Some authors have found few infected fish, if any, in the areas they have examined (Puerto Rico: Bunkley-Williams and Williams, 1994; southern California: Wilkie and Gordin, 1969) . Others have found that low levels of infection are not uncommon (e.g. southern Queensland; Diggles and Lester, 1996c). Keeping multiple fish in holding tanks and at aquarium stores increases the chances of a fish carrying "Ich" parasites, but it is still possible to acquire a fish that is not infected with "Ich".
If new fish are quarantined for at least 6 weeks, any parasites on the fish will have gone through a number of life cycles increasing the number of parasites present. In the majority of cases, the increase in parasite numbers will result in full blown infection and fish can be treated to remove the parasites. Hyposalinity has been demonstrated to break the life cycle of "Ich" (Cheung et al. 1979; Colorni, 1985) and fish correctly treated with hyposalinity will be free from "Ich". Any fish that do not show signs of infection after 6 weeks are very unlikely to be carrying any parasites.
If fish that are free from "Ich" (either because they were not originally infected or because they have been treated with hyposalinity) are added to an aquarium that is free from "Ich", the aquarium will stay free from "Ich" and be another exception to "Ich" always being present.
Burgess and Matthews (1994) were attempting to maintain a viable population of C. irritans which could be used in later studies. To maintain the parasite populations, they needed host fish in order for the trophonts to feed and continue the life cycle. Each host fish was only used once in a process of serial transition such that none of the hosts would die or develop an immunity. While the procedure worked very well and enabled them to maintain populations for some time, the viability of the populations decreased with time and none of the 7 isolates they used survived more than 34 cycles, around 10 to 11 months. They suggest this is due to senescence and aging in cell lines is well recognised in Ciliophora.
The presence of aging cell lines in C. irritans suggests that an aquarium that has been running for longer than 12 months without any additions is unlikely to have any surviving "Ich" parasites, yet another exception to "Ich" always being present.
Whilst "Ich" may be present in some aquaria, it is certainly not present in all aquaria. Through careful quarantining and treatment, it is very much possible to establish and maintain an "Ich" free aquarium.
Stress and poor water conditions do not cause marine "Ich", although they will lower a fish's resistance to infection and impair their immune system. If C. irritans is not present in a tank, it doesn't matter what how stressed a fish may be, it cannot get infected. In a tank where parasites are present, stressed fish are more likely to show signs of "Ich" before more healthy fish, but the healthy fish are just as likely to become infected as the numbers of parasites increase. Those fish species that are less susceptible to "Ich" or those individuals that have an acquired immunity, may show no signs and may not get infected.
One common argument against the use of natural seawater is the possibility of the introduction of parasites. While it is theoretically possible to introduce C. irritans, it is practically very unlikely owing to the nature of the life cycle of the parasite.
As discussed above, C. irritans spends very little time in the water column. After dropping off the host fish, trophonts head straight to the substrate to reproduce. This may take as little as 30 minutes but could extend to 24 hours (Cheung et al., 1979). Burgess and Matthews (1994b) found that significantly more trophonts left their host during darkness while fish are resting. This would greatly decrease the chances of trophonts being swept away from the substrate. These two factors combined almost rule out the possibility of trophonts being collected with natural seawater.
Excystment of theronts from tomonts also happens at night (Burgess and Matthews (1994b) and as theronts are only viable for a few hours, the chances of collecting theronts is low and those collected will most likely die before use in an aquarium.
Bunkley-Williams L. and Williams E.H. 1994. Disease caused by Trichodina spheroidesi and Cryptocaryon irritans (Ciliophora) in wild coral reef fishes. Journal of Aquatic Animal Health 6:360-361.
Burgess P.J. and Matthews R.A. 1994. A standardized method for the in vivo maintenance of Cryptocaryon irritans (Ciliophora) using the grey mullet Chelon labrosus as an experimental host. J Parasitol 80:288-292.
Burgess P.J. and Matthews R.A. 1994. Cryotocaryon irritans (Ciliophora): photoperiod and transmission in marine fish. Journal of the Marine Biological Association of the United Kingdom 74:535-542.
Burgess P.J. and Matthews R.A. 1995. Cryptocaryon irritans (Ciliophora): acquired protective immunity in the thick-lipped mullet, Chelon labrosus. Fish & Shellfish Immunology 5(6):459-468.
Cheung P.J., Nigrelli R.F. and Ruggieri G.D. 1979. Studies on cryptocaryoniasis in marine fish: effect of temperature and salinity on reproductive cycle of Cryptocaryon irritans Brown, 1951. J. Fish Dis. 2:93-97.
Colorni A. 1985. Aspects of the biology of Cryptocaryon irritans, and hyposalinity as a control measure in cultured gilt-head sea bream Sparus aurata. Dis. Aquat. Org. 1:19-22.
Colorni A. 1987. Biology of Cryptocaryon irritans and strategies for its control. Aquaculture 67(1-2):236-237.
Delbeek J.C. and Sprung J. 1994. The Reef Aquarium Volume 1. Ricordea Publishing, Coconut Grove, Florida. 544pp.
Dickerson H.W. and Clark T.G. 1996. Immune response of fishes to ciliates. Annual Review of Fish Diseases 6:107-120.
Dickerson H.W. and Dawe D.L. 1995. Icthyophythirius multifiliis and Cryptocaryon irritans (Phylum Ciliophora) In: P T K Woo (ed.) Fish Diseases and Disorders. Volume 1: Protozoan and Metazoan Infections. CAB International, Wallingford, Oxon. pp 181-227.
Diggles B.K. and Lester R.J. 1996. Influence of temperature and host species on the development of Cryptocaryon irritans. J Parasitol 82:45-51.
Diggles B.K. and Lester R.J. 1996. Variation in the development of two isolates of Cryptocaryon irritans. J Parasitol 82:384-388.
Grutter A.S. 2000. Ontogenetic variation in the diet of the cleaner fish Labroides dimidiatus and its ecological consequences. Mar. Ecol. Prog. Ser. 197:241-246.
Ruppert E.E. and Barnes R.D. 1994. Invertebrate Zoology. Saunders College Publishing, Orlando, FL. 1056pp.
Yoshinaga T. and Dickerson H.W. 1994. Laboratory propagation of Cryptocaryon irritans Brown, 1951 on saltwater-adapted black mollies Poecilia latipinna. J. Aquat. Anim. Health 6:197-201.
Wilkie D.W. and Gordin H. 1969. Outbreak of cryptocaryoniasis in marine aquaria at Scripps Institution of Oceanography. California Fish and Game 55:227-236.
Wright A. and Colorni A. 2002. Taxonomic re-assignment of Cryptocaryon irritans , a marine fish parasite.. European Journal of Protistology 37(4):375-378.
Last updated: March 26, 2006